Making Sense of Social Media Conversations
The relevance of social media monitoring and engagement in the healthcare industry
Billions of conversations take place every month on social media platforms including Twitter, Facebook, Instagram, as well as blogs and forums. These conversations are not only taking place in social settings, but are influencing companies as well. Social media use continues to rise as we see more people using it in both their personal and professional lives — and brands are responding by continually looking for better ways to engage with them. All of these conversations comprise pools of rich data, but listening to them for meaning can be challenging. So, how do companies make sense of all this endless data to determine how and where to listen, identify what consumers are talking about, classify the types of content they are posting and understand the behaviors they are engaging in?
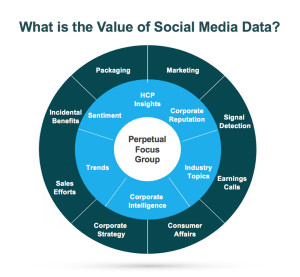 In the healthcare industry, patients and healthcare providers (HCPs) are looking to social media for information and support regarding their health and the health of their patients. They serve as a “perpetual focus group,” whose conversations taking place on social media provide brands with the opportunity to listen. Patients share their opinions, ask questions about diseases and treatment options, and directly or incidentally report adverse events. Meanwhile, HCPs voice opinions on the healthcare industry, provide thought leadership on a wide range of topics such as participatory medicine, reimbursement, and medical education. In a recent survey, more than 40 percent of respondents reported that information found via social media would affect the way they coped with a chronic condition or their approach to diet and exercise; 34 percent said it would affect taking certain medication.1 This demonstrates to companies that patients are directly impacted by what they view on social media and in order for brands to leverage that data radiating out of all the social conversations, several factors need to be in place.
In the healthcare industry, patients and healthcare providers (HCPs) are looking to social media for information and support regarding their health and the health of their patients. They serve as a “perpetual focus group,” whose conversations taking place on social media provide brands with the opportunity to listen. Patients share their opinions, ask questions about diseases and treatment options, and directly or incidentally report adverse events. Meanwhile, HCPs voice opinions on the healthcare industry, provide thought leadership on a wide range of topics such as participatory medicine, reimbursement, and medical education. In a recent survey, more than 40 percent of respondents reported that information found via social media would affect the way they coped with a chronic condition or their approach to diet and exercise; 34 percent said it would affect taking certain medication.1 This demonstrates to companies that patients are directly impacted by what they view on social media and in order for brands to leverage that data radiating out of all the social conversations, several factors need to be in place.
When determining how to utilize the data in social media conversations, one rule-of-thumb is to work backward — start with objectives and determine which categories of data would support the analysis and provide actionable insights. Companies can build a tagging structure based on relevant categories, such as: source (e.g., social media platform), author, type, topic, behavior, and sentiment. For example, if a bran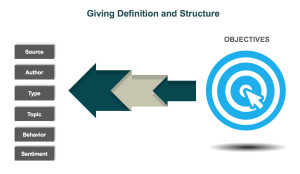 d is looking to identify who is talking about a product, the kinds of content they are posting, and the topics within their content, a structure that would classify posts into categories such as Consumer, Branded Product, and Medical Inquiry could be utilized. As more and more posts are tagged, patterns, trends, and themes can be identified.
d is looking to identify who is talking about a product, the kinds of content they are posting, and the topics within their content, a structure that would classify posts into categories such as Consumer, Branded Product, and Medical Inquiry could be utilized. As more and more posts are tagged, patterns, trends, and themes can be identified.
Social media strategists must be cognizant of the regulations pharmaceutical companies face when approaching social media usage and determining involvement. Many pharmaceutical companies have legitimate concerns regarding social media participation, including the need to have:
- Identification, triaging, and reporting of Adverse Events (AEs), Product Quality Complaints (PQCs)
- Consistency of messaging and having a team that can adhere to social media promotion guidelines and regulations
- Previous experience and internal knowledge in terms of social media engagement and usage
The current regulatory environment is understandably daunting and serious consideration must be applied when developing a social strategy. While these challenges exist today and are likely to remain for the foreseeable future, there are prescriptive solutions and industry best practices available to mitigate risk. Regulations and the strategies that come about as a result can impact a pharmaceutical company’s ability to continue that social media conversation. Reliable processes can be implemented, borrowing best practices from existing patient interaction policies, as well as social monitoring and engagement strategies from other regulated industries.
Other key steps and factors to consider to successfully make sense of social conversations include:
- Train staff – Reduce risk by utilizing highly trained specialists experienced in the nuance of monitoring, identifying, and triaging AEs.
- Establish documented workflows – Configure a systematic approach to the identification of AEs and PQCs and their triaging to the appropriate Pharmacovigilance point-of-intake.
- Technology – Utilize cutting-edge technology platforms to monitor and tag conversations. Although there is no replacement for human engagement, as volume scales, technology becomes increasingly critical for success.
The importance of listening is always stressed in the pharmaceutical industry, but the actual work requires an appreciation of how different people “speak” on social media (patients versus HCPs), understanding the value of categorical organization of data, and the selection and application of appropriate technology. There is no silver bullet, but building meaningful structures out of disparate sets of data is doable – in fact, it’s necessary if the industry is to make the most out of today’s social media landscape. Leveraging social media data and engagement opportunities are crucial imperatives for companies to support key business goals focused around patient and provider programs — and it will pay off.
Reference
- Social Media ‘Likes’ Healthcare: From Marketing to Social Business. PWC Health Research Institute. April 2012

